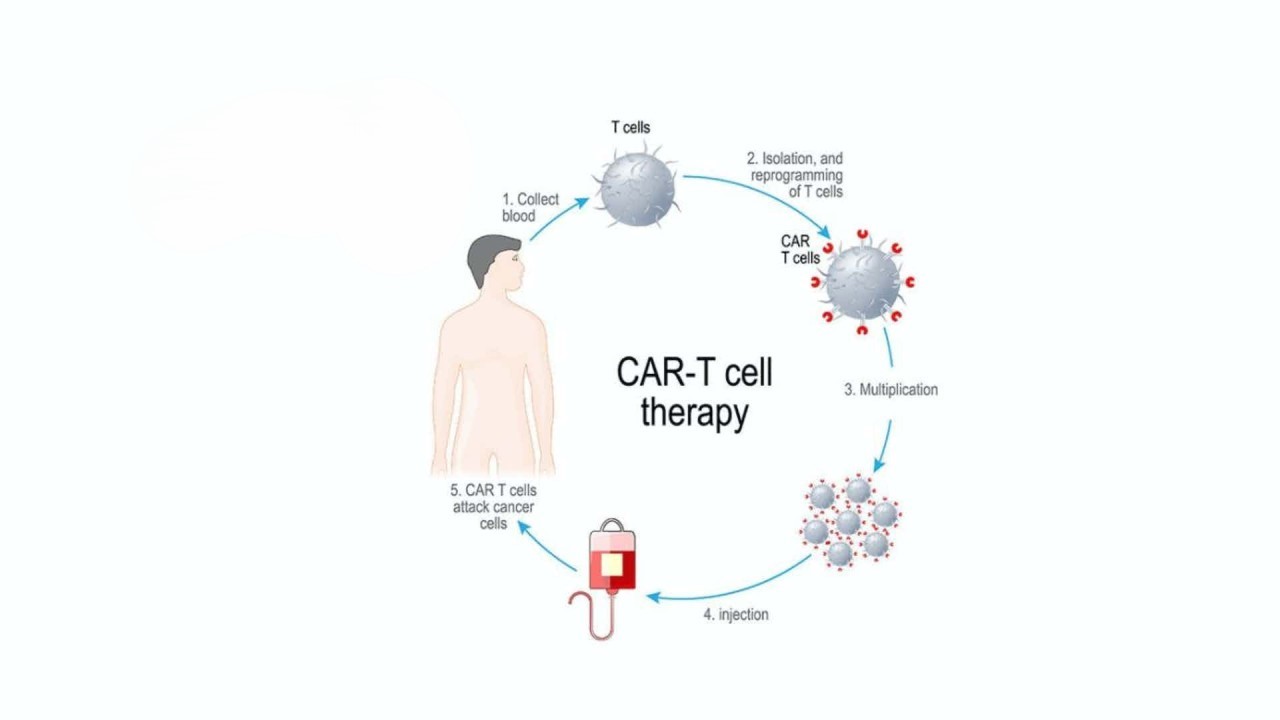
T cells are a component of the immune system that the body uses to combat cancer and other illnesses. Genetically altered T cells are known as chimeric antigen receptor T (CAR T) cells. Using the ability of altered T cells to locate and eradicate cancer cells, CAR T cell therapy improves the body's natural barriers against the illness.
Like blood donation, blood is collected from the patients to separate T cells. These T cells are then modified in the laboratory under expert supervision to express the Chimeric Antigen Receptor. Before Car T cell administration, patients need to undergo chemotherapy to prepare their bodies for Car T cell therapy. Once infused, Car T cells are anticipated to recognize and neutralize resistant cancer cells by activating the body’s immune system.
Since 2017, the FDA has approved six Car T cell therapies for treating blood cancers like Lymphomas, certain types of Leukemia and most recently, multiple myeloma. Patients eligible for Car T cell therapy must meet FDA criteria and be in overall good health to handle the waiting period and potential side effects.
